Allergy
In Australia, allergic disease is the fastest growing chronic medical condition. Most current treatment options for allergic disease provide symptomatic relief only and are inadequate for severe allergic disorders.
“Allergen immunotherapy involves the regular administration of gradually increasing doses of allergen extracts over a period of years. Immunotherapy can be given to patients as an injection or as drops or tablets under the tongue (sublingual). Allergen immunotherapy changes the way the immune system reacts to allergens, by switching off allergy. The end result is that you become immune to the allergens, so that you can tolerate them with fewer or no symptoms” (Australiasian Society of Clinical Immunology and Allergy).
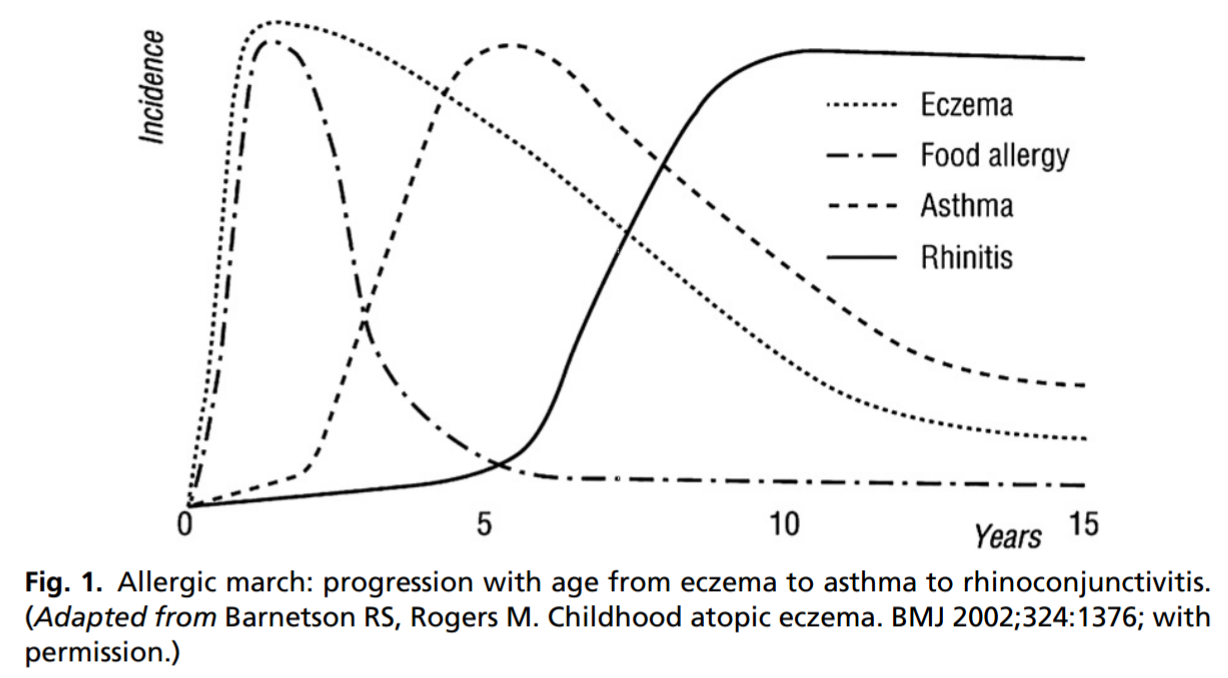
The term “Allergic March” refers to the progression of atopic diseases as a child grows. Eczema generally presents in the first months of life in atopic individuals, along with sensitisation to food proteins, for example proteins in hen’s eggs and cow’s milk. Asthma has a later onset, followed by atopic rhinitis (Fig. 1) . Allergen immunotherapy plays a potential role in interrupting the allergic march and preventing the development of new atopic diseases in children with already established allergic symptoms.
Native Allergen vs. Allergoid
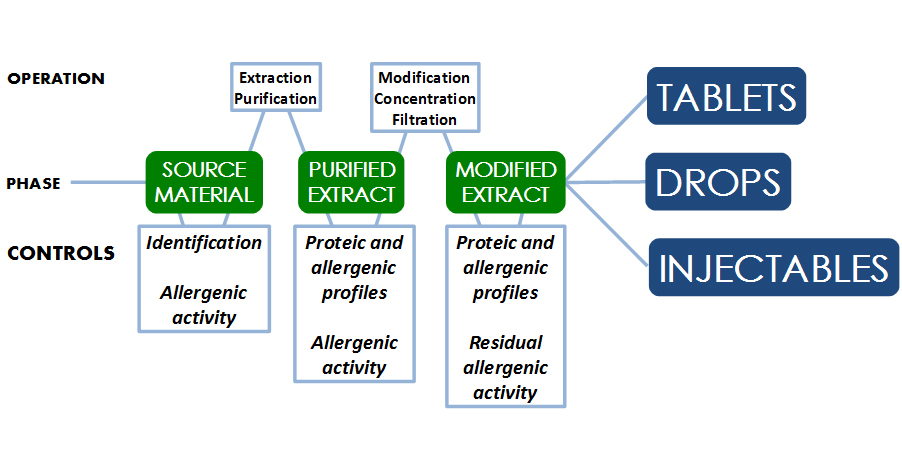
Allergen proteins used in immunotherapy fall into two main groups: native allergens and chemically-modified allergens called "allergoids". A native allergen is an allergenic compound or protein derived from natural sources. Its molecular linkage and three-dimensional structure remain unchanged after the extraction and purification process. An allergoid is a chemically-modified native allergen and has modified molecular linkage and three-dimensional structure. Recombinant allergens have become the new trend for the future of allergen immunotherapy; however, there are currently no commercial recombinant allergen vaccines currently available in the market.
Why Allergoid?
In allergen immunotherapy, the native allergen contains both T-cell epitopes recognized by the MHC-TCR system and reaginic IgE epitopes, which are responsible for T-cell presentation and IgE binding, respectively. T-cell presentation is related to immunogenic activity, which contributes to the efficacy of immunotherapy.
On the other hand, IgE binding is related to undesirable adverse reactions of immunotherapy. In the case of sublingual administration, the native allergen also reacts with protease in the gastrointestinal tract after being swallowed, which causes the damage and degradation of the allergen. The ingested allergen is further destroyed in the stomach and in the bowel by enzymes, reducing the antigenicity of allergen SLIT (Sublingual Immunotherapy).
Two Different Allergoids
Based on different chemical reactions, allergoids are classified into two different types:
1) Polymeric Allergoid: the native allergen is reacted with aldehydes (glutaraldehyde, formaldehyde) to form highly cross-linked protein aggregates. During this polymerization process, the molecular weight and structure of the allergens become much larger, while IgE epitopes remain hidden inside the polymeric structure.
The re-arrangement of the allergen structure reduces IgE binding and may impair T-cell recognition. This means that the efficacy of immunotherapy could be in part achieved with improved side effects. In addition, the introduction of new determinants by chemical modification can be excluded. Polymeric allergoids are used in SCIT (Subcutanenous Immunotherapy) due to its large molecular size, which will likely cause low penetration via the oral mucosa.
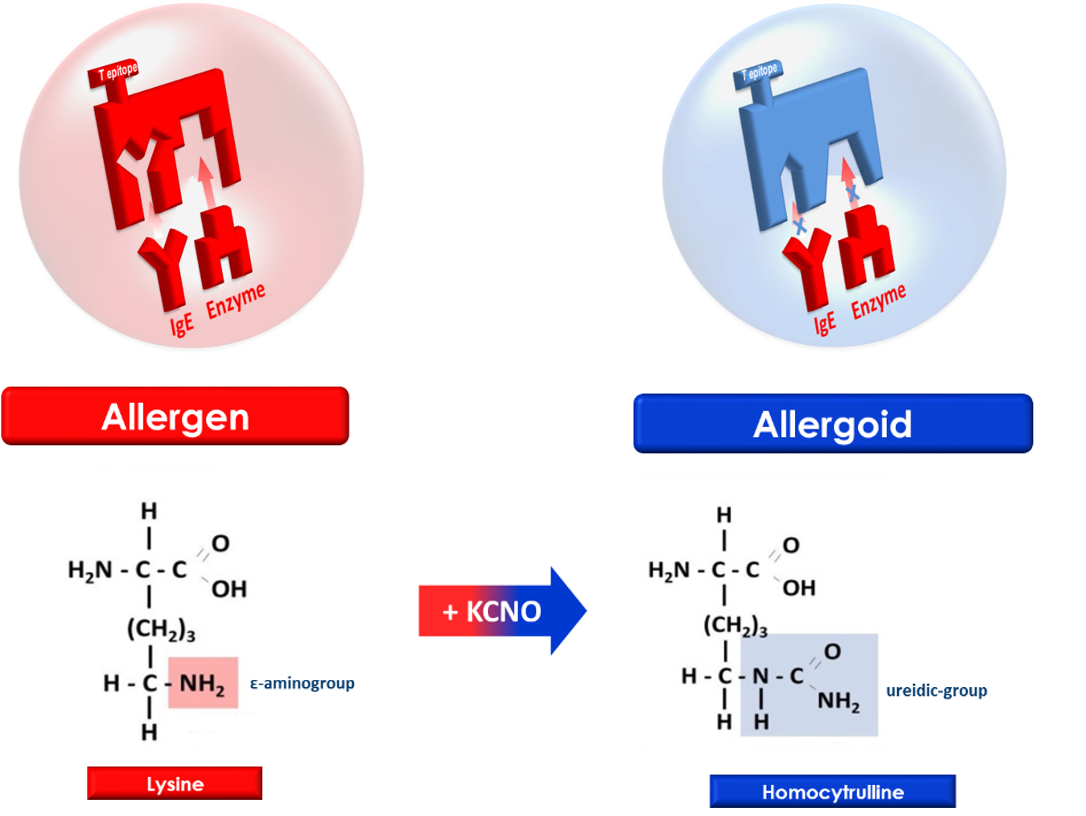
2) Monomeric Allergoid: the monomeric carbamylated allergoid is obtained by selective carbamylation with potassium cyanate at alkaline pH, a reaction that leads to a marked transformation of є-aminogroups of allergen lysine residues, critical for specific IgE affinity and proteolysis of enzymes.
References:
1. Australasian Society of Clinical Immunology and Allergy 2014, Immunotherapy for Treatment of Allergy, Australasian Society of Clinical Immunology, Australia.
2. Gordon, B. 2011, ‘The Allergic March: Can We Prevent Allergies and Asthma?’, Otolaryngologic Clinics of North America, vol. 44, issue 3, pp. 765-777.
